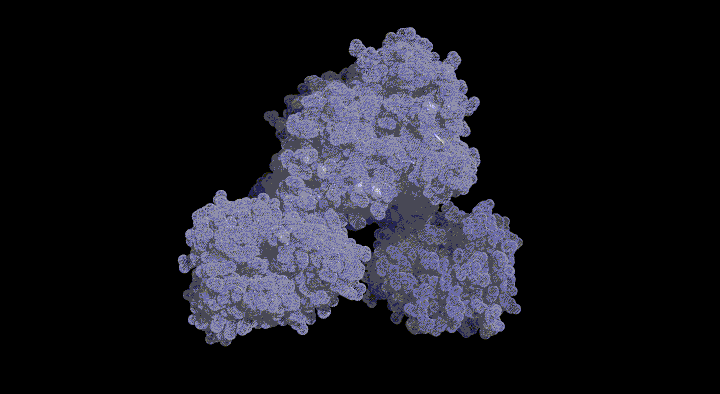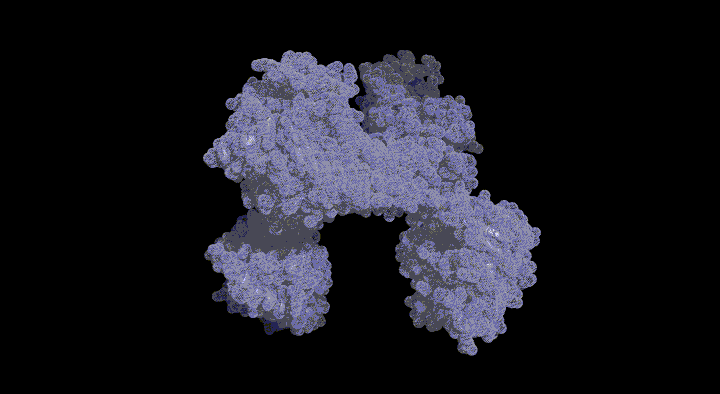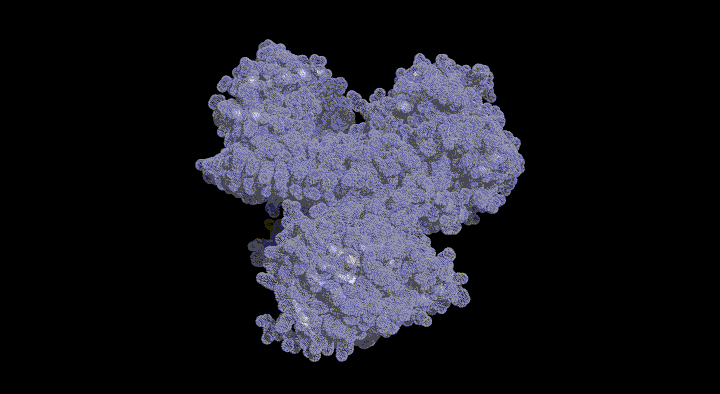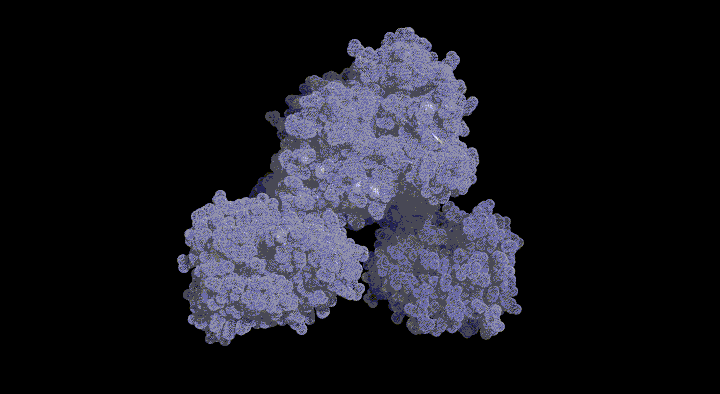
Glutathione Degrading Enzymes
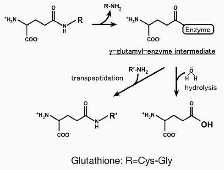
Glutathione (GSH; γ-Glu-Cys-Gly), an essential antioxidant for living organisms, possesses a specific peptide bond via the glutamate side chain γ-glutamyl linkage)
and is resistant to degradation by general proteases.
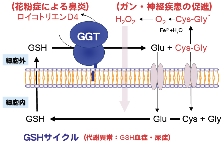
γ-Glutamyltranspeptidase (GGT) is the only enzyme capable of cleaving the γ-glutamyl linkage of GSH. As a key enzyme responsible for the initial steps of GSH metabolism and xenobiotic detoxification, GGT plays a decisive role in the defense against oxidative stress. Furthermore, it is deeply implicated in a wide range of diseases, including arteriosclerosis, renal impairment, Parkinson's disease, and multidrug resistance in cancer cells.
GGT belongs to the N-terminal nucleophile (Ntn) hydrolase family; its inactive precursor matures into an active heterodimer through autocatalytic processing.
In this study, we aim to elucidate the molecular mechanisms of GGT by structurally tracking the entire pathway from its maturation process to its reaction mechanism.
 .
. 
Elucidation of the Reaction Mechanism of GGT
[Proc. Natl. Acad. Sci. USA, 103, 6471–6476 (2006)]
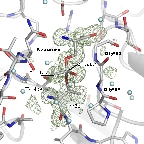
Although this enzyme was discovered in the 1960s, its reaction mechanism remained shrouded in mystery for many years. We succeeded in determining the structure of the mature form of Escherichia coli-derived GGT. Furthermore, by utilizing flash-cooling techniques, we trapped the acyl-enzyme intermediate (where the γ-glutamyl group is bound to the N-terminal Thr391 of the small subunit, which serves as the active residue) and elucidated its structure.
These structures, along with the GGT-glutamate complex structure, provided the structural basis for understanding substrate recognition, the molecular mechanism of the reaction, and mutational analysis.
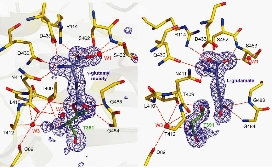
Elucidation of the Autocatalytic Maturation Mechanism of GGT [J. Biol. Chem., 288, 2433–2439 (2007)]
GGT, a key enzyme in glutathione metabolism, is synthesized from a single open reading frame. It undergoes post-translational processing to be cleaved into two subunits, forming a heterodimer to become the mature enzyme. Biochemical experiments had previously established that this reaction occurs autocatalytically and that Thr-391 serves as the catalytic residue for both the processing event and the enzymatic reaction.
We determined the structure of the T391A mutant, which mimics the precursor state, and elucidated the process of autocatalytic maturation. Structural comparison with the mature enzyme revealed that the precursor GGT undergoes conformational changes upon processing. This processing is essential for the formation of the substrate-binding pocket, thereby enabling specific substrate recognition.
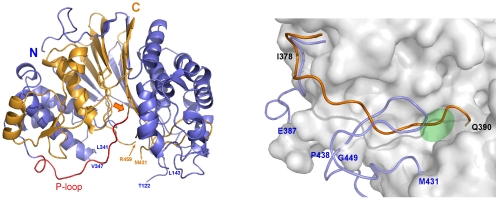
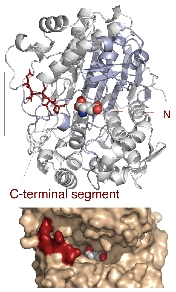
Crystal Structure of Precursor GGT The orange arrow indicates the cleavage site of autocatalytic processing.
Structural Changes of GGT upon Maturation The structure of the precursor GGT is shown in orange, and the mature GGT is shown in blue. The active site, indicated in green, is formed by the structural changes resulting from processing.
Reaction Mode of GGT Inhibitors
[J. Mol. Biol., 380, 361-372 (2008)]
Human blood GGT activity ($\gamma$-GTP level) is widely used as a diagnostic marker because it increases significantly in hepatobiliary diseases such as liver cancer and alcoholic liver disease. Recently, associations between GGT activity and the onset of conditions including Parkinson's disease, cardiovascular diseases, and neurodegenerative disorders have been reported. Consequently, controlling the activity of this enzyme is attracting increasing attention from a medical perspective.
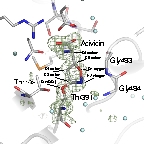
In this study, we determined the crystal structures of Escherichia coli GGT in complex with two substrate analogue inhibitors, azaserine and acivicin. These structures elucidated the detailed binding sites and binding modes of these inhibitors, which had previously remained unclear. Both inhibitors were found to bind to the substrate pocket by forming a covalent bond with the catalytic residue of GGT (Thr391).
Interestingly, in the GGT-azaserine complex, the carbon atom attacked by Thr391 adopted a tetrahedral configuration, indicating that the transition state of the reaction was stabilized. Similarly, acivicin, which possesses a five-membered ring structure, also adopted a tetrahedral configuration (bonded via $sp^3$ hybrid orbitals) with Thr391. This suggested a novel reaction mechanism for the covalent modification by acivicin, involving the opening and closing of the five-membered ring.
Based on the similarities in amino acid sequences and reaction types, this inhibition mechanism is suggested to be common not only to E. coli GGT but also to the superfamily of glutamine (glutamate)-utilizing enzymes (GGT and Gat families) found in organisms ranging from bacteria to mammals.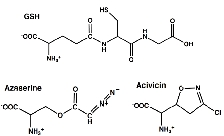
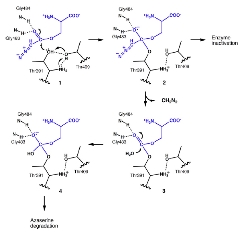
Reaction Mechanism of the GGT Inhibitor Azaserin
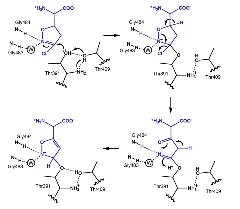
Reaction Mechanism of Acivicin Acivicin covalently binds to GGT through a reaction involving the opening and closing of its ring structure, rather than via a simple nucleophilic substitution reaction.
Function of the Lid-Loop Covering the Substrate Pocket
[FEBS J., 277, 100-1009 (2010)]
The catalytic pocket of GGT contains a loop (lid-loop) that covers the substrate-derived $\gamma$-glutamyl group. It has been revealed that this loop is disordered when the catalytic pocket is empty, but becomes fixed to cover the pocket in the presence of a substrate. Since the introduction of mutations into amino acids on the lid-loop significantly reduced enzymatic activity, we hypothesized that this loop was essential for the GGT enzymatic reaction and involved in substrate recruitment.
Surprisingly, however, several species possess GGTs that lack the lid-loop sequence. To elucidate how such GGTs recognize substrates, we first attempted the crystal structure analysis of Bacillus subtilis GGT.
The structure of the B. subtilis GGT–substrate analog complex revealed no loop covering the catalytic pocket; instead, the bound substrate analog was largely exposed to the solvent. The acquisition of this structure overturned the prevailing concept regarding the role of the lid-loop in the enzymatic reaction. We plan to investigate the details further through mutagenesis and in vivo studies.
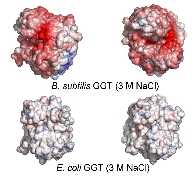
Furthermore, B. subtilis GGT is a salt-tolerant enzyme capable of retaining activity even in the presence of 3M NaCl, and its application in the brewing industry is anticipated (collaborators are currently conducting applied research). Calculations of molecular surface electrostatic potential suggested that even under high-salt conditions where E. coli GGT loses its surface charge, B. subtilis GGT remains strongly negatively charged. We believe this is a contributing factor to its salt tolerance.